Durham maker of a medical resuscitation device raises $8 million from investors
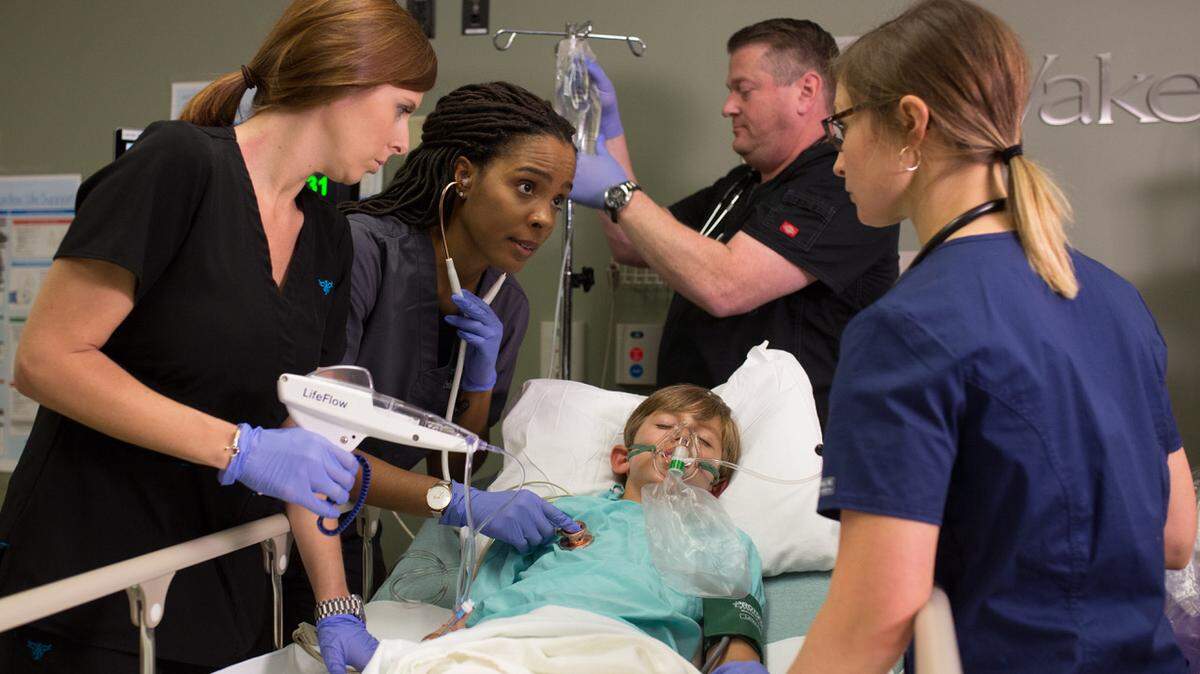
Durham-based medical device maker 410 Medical has raised a new $8 million round of funding — an influx of cash that will help the young company expand its efforts to increase sales of its device called LifeFlow.
LifeFlow is used in resuscitating critically ill patients and was founded by a doctor at WakeMed in Raleigh in 2013.
Since then the company has raised more than $18 million from investors, including several health care networks, such as WakeMed. The company now operates out of an office in the American Underground in downtown Durham, where around 10 of its employees work. In total, the company has 14 full-time employees.
The latest round of funding was led by Durham-based investment group Hatteras Venture Partners, which specializes in biomedical companies, as well as the investment arm of OSF Healthcare, an Illinois-based health system that has 13 hospitals.
“It’s an exciting validation of the effectiveness of the product,” 410 Medical CEO Kyle Chenet said about the new investment. “OSF is an investor and user, which gives a lot of validation.”
LifeFlow, which is manufactured in Franklin County, received clearance from the U.S. Food and Drug Administration for human use in 2016.
Since then, the company’s device has been sold to 35 different hospital networks and has been used on thousands of patients, Chenet told The News & Observer. Hospitals like WakeMed, Cincinnati Children’s Hospital Medical Center and Children’s Hospital Colorado use the device.
LifeFlow, the gun-shaped, hand-held device, helps clinicians deliver fluids quickly and efficiently to patients with life-threatening conditions, like shock, sepsis and trauma, who are are at a higher risk for organ failure.
Chenet added that Hatteras, whose partner Jeff Terrell will join the board of the company, provides experience when it comes to commercializing LifeFlow.
Chenet said the company plans to use the money to fund some additional clinical data on its device and to boost its sales and marketing staff. Having more clinical data, he noted, will help in its sales efforts to hospitals.
“We have been fairly lean [in staffing], and we will continue to be lean,” he said. But the company will need more employees as it puts product in more hospitals, especially as it plans to unveil a second product next year.
Chenet wouldn’t provide details about the second product, beyond saying it will be focused on resuscitation as well.
Earlier this year, the company introduced a new version of LifeFlow.
Chenet said that the latest iteration of the device can deliver fluids to patients up to 10 times faster than traditional methods.
410 Medical was co-founded by Dr. Mark Piehl, a pediatric intensivist at WakeMed in Raleigh. Piehl is a graduate of the UNC-Chapel Hill Medical School.
Other investors include the angel investment networks at all three of the Triangle’s largest universities: Duke Angel Network, Carolina Angel Network and Wolfpack Investor Network.
This story was produced with financial support from a coalition of partners led by Innovate Raleigh as part of an independent journalism fellowship program. The N&O maintains full editorial control of the work. Learn more; go to bit.ly/newsinnovate
This story was originally published October 23, 2019 at 10:07 AM.