‘Particulate matter,’ silica and iron oxide pieces, in cancer drug causes recall
One lot of an injection drug meant to help patients deal with leukemia has been recalled on concerns it might damage or kill the patient.
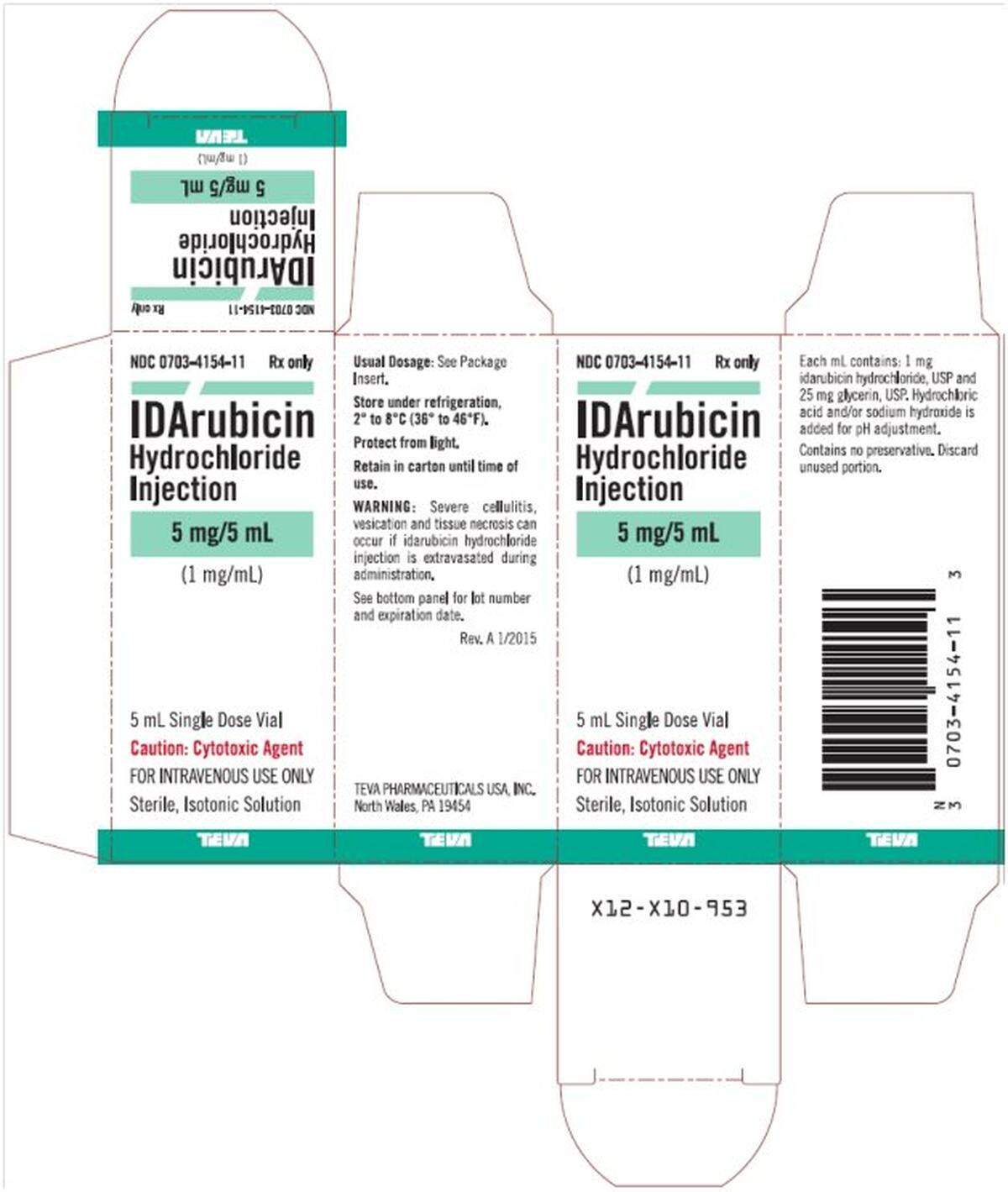
That’s not overstatement, but, rather, the summary of the reason Teva Pharmaceuticals recalled one lot of IDArubicin Hydrochloride Injection 5 mg/5 ml vials.
Teva’s FDA-posted recall notice says the recall “is initiated based on an internal inspection that found particulate matter in one vial of the product identified as silica and iron oxide.”
Injecting something “that contains particulate matter may result in local irritation or swelling in response to the foreign material,” the alert says. “If the particulate matter reaches the blood vessels it can travel to various organs and block blood vessels in the heart, lungs or brain which can cause stroke and even lead to death.”
Teva also says “the likelihood of patient harm is remote or unlikely,” but the action of a recall speaks loudly.
What you should do now if you have recalled vials
Lot No. 31329657B, expiration 08/2023, has been recalled. First, talk to your medical professional. Then, return the vials.
Teva claims it has notified customers, but if you want instructions on returning it or have any questions related to the recall, call 888-838-2872, and hit option No. 4, Monday through Friday, 9 a.m. to 5 p.m., Eastern time. Voice mail should take calls outside of those hours.
If this or any drug causes a problem, after notifying a medical professional, let the FDA know via its MedWatch Adverse Event page or by filling out a form you can get by calling 800-332-1088.
After that, let the company know. In this case, that would be Teva at 888-838-2872, option 3, then, option 4, Monday through Friday, 9 a.m. to 5 p.m., Eastern time, or email druginfo@tevapharm.com.
This story was originally published March 30, 2022 at 10:13 AM with the headline "‘Particulate matter,’ silica and iron oxide pieces, in cancer drug causes recall."
